Gene therapy is a highly promising field that has nevertheless suffered from various setbacks since it was initially proposed as a viable treatment for an array of diseases. Despite this, the progress made in recent decades has been astounding.
To test the use of the protein CCN5 to treat cardiac fibrosis in large animals and understand the molecular mechanisms involved. To carry out trials using mouse models to investigate the efficacy of using a plant viral protease, NIa, as a gene therapy to treat age-related macular degeneration and Alzheimer’s disease, with the ultimate goal of translating these findings into humans
 CCN5 Secreted protein
CCN5 Secreted protein
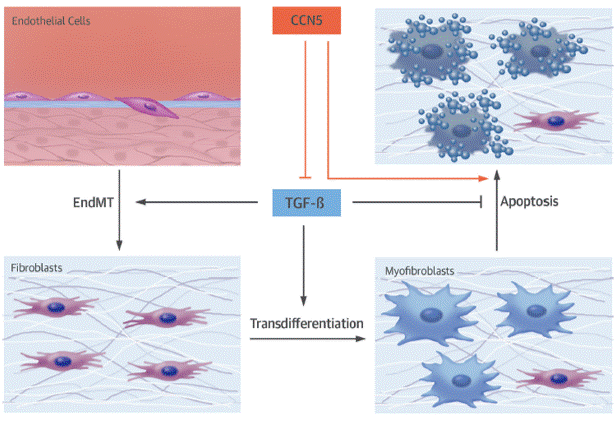
Cardiac fibrosis is characterized by replacement of cardiac cells with fibrous connective tissue, causing scarring and resulting in a stiffer and less compliant cardiac muscle. It is an independent predictor for heart failure progression and is a valid target for treatment. Heart failure accounts for approximately 450,000 deaths per year in the United States. Our team found that expression of a matricellular protein, CCN5, is significantly reduced in the myocardium of patients with severe heart failure and, more importantly, that CCN5 can reverse established cardiac fibrosis in animal models of heart failure.
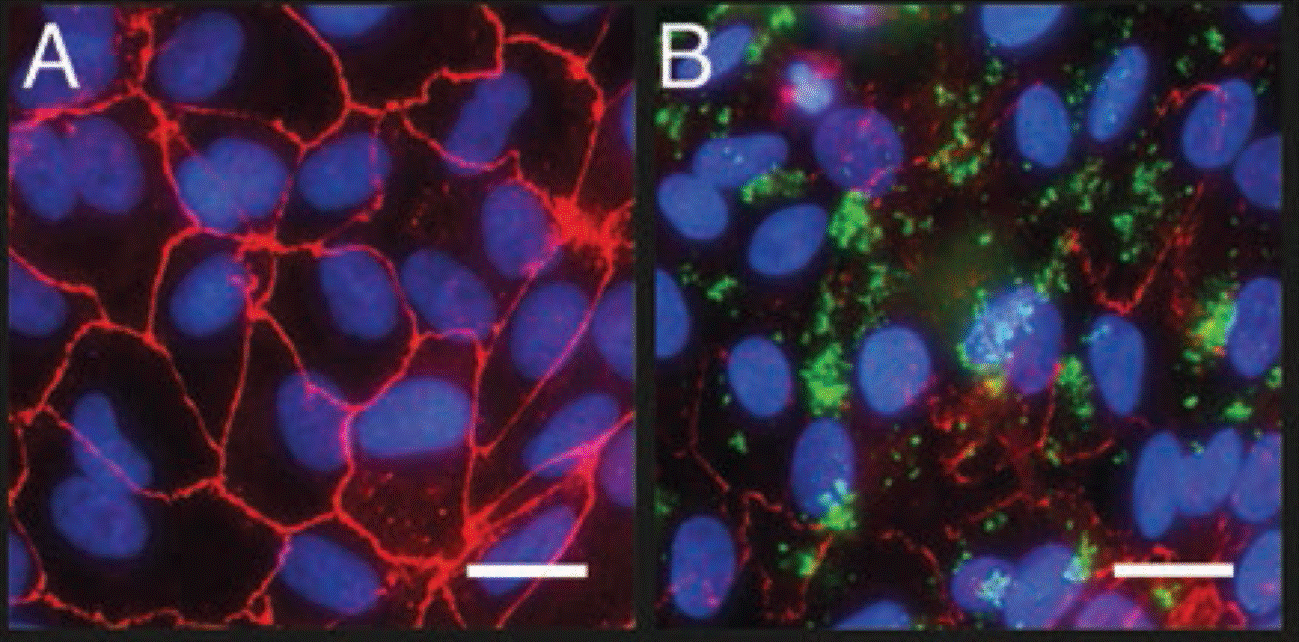
Our team showed that CCN5 inhibits cardiac fibrosis by blocking endothelial-mesenchymal transition and transdifferentiation of fibroblasts in the heart. In addition, CCN5 reverses established cardiac fibrosis by inducing apoptosis in myofibroblasts but not in myocytes or fibroblasts. The anti-fibrotic activity of CCN5 was further confirmed in sheep and pig models of heart failure. Cardiac fibrosis is implicated in the pathogenesis of diverse heart diseases, including heart failure with preserved ejection fraction (HFpEF), atrial fibrillation, and muscular dystrophy. The first line target of CCN5 will be HFpEF, followed by atrial fibrillation and muscular dystrophy.
 NIa Protease
NIa Protease
ALZHEIMER’S DISEASE IS the most common type of dementia – a condition resulting in the loss of mental ability associated with the gradual death of brain cells. The number of people living with dementia worldwide is currently estimated at 44.4 million. Affecting memory, thinking, behaviour and the ability to perform everyday activities, the disease is expected to affect an estimated 135.5 million people by 2050, and as such is in dire need of effective therapeutics. Dementia is one of the most devastating illnesses for both sufferers and their families, as patients lose their memories, independence, relationships and ultimately their lives. Furthermore, there is already a huge fiscal burden on the healthcare service, which will only increase with an ageing population. Despite its prevalence, and the decades of research that scientists have invested, a cure for Alzheimer’s disease still remains elusive.
Understanding the factors underlying a disease’s development is often the first step towards discovering a potential treatment. Until now, therapies for Alzheimer’s disease have been ultimately unsuccessful as they have treated the symptoms, rather than the root cause. The next generation of treatments will need to tackle the cell death in the brain that underlies this condition. The primary culprit of Alzheimer’s disease is a small peptide called amyloid beta. The peptide exists as three different forms in the human brain: a monomer, an oligomer, and also in aggregated plaques. Of the three, the oligomeric amyloid beta (OAβ) peptide is known to be the most toxic. When accumulated in the neuronal cells, OAβ causes malfunction and ultimately leads to the death of these cells and shrinkage of the brain.
NIa is a naturally occurring plant viral protease that has the ability to cleave OAβ. The phenotypes that the Alzheimer’s mice possess, including both anatomical characteristics such as increased amyloid beta plaques and behavioural deficits such as impaired memory or increased anxiety, were significantly ameliorated following the NIa gene transfer.